|
Procedures |
|
| Procedures Contact Details |
The Generalised Process
This stage is vital as it removes any waste food products that may be in the vegetable oil, it is also useful to remove any excess water that may be in the oil at this stage.
The amount of alcohol is fairly fixed throughout all processes; the amount of catalyst depends upon the alcohol used, the catalyst used and the oil used in the procedure. See the experimental section of this website for further details.
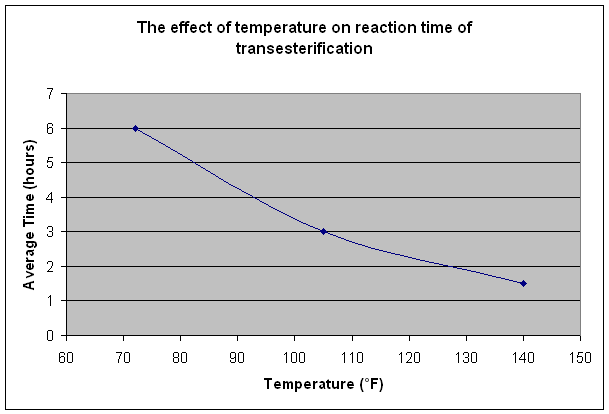
Transesterification is the process of exchanging the alkoxy group of an ester compound by another alcohol. These reactions are often catalysed by the addition of an acid or base. It has been found that a vigorous stirring at the initial stage is necessary to bring the oil, catalyst and alcohol into intimate contact, however towards the end of the reaction it can be beneficial to the reaction to decrease mixing. Although it is advertised that heating is necessary at the reaction process this is not specifically correct. Heating during this process just speeds up the reaction as is shown in the graph below (data from ‘Biodiesel Production Technology, J H Van Gerpen, Biological and Agricultural Engineering, June 2006):
Although it may seem like a great idea to increase the temperature so that the reaction takes less time the boiling point of the alcohol has to be taken into account (methanol ~65oC, ethanol ~78oC)
This allows the glycerol, excess alcohol and other waste products and impurities separate from the biodiesel. The biodiesel will end up on top of the waste product.
The biodiesel has to be separated from the glycerol by-product which can be further processed or disposed of in the correct manor (see by-products section).
The washing process is designed to purify the biodiesel further. There are various procedures that can be taken, bubble washing, mist washing and ‘dry’ washing are a few examples. There are legal requirements that accompany washing the product with water (see legal requirements section). Most of the impurities are in the glycerine, though there are still small amounts of lye, soap, ethanol and water in the biodiesel, which will cause trouble in your fuel system and engine. An easy way to remove them is by washing the fuel with water. Add water to the biodiesel until you have about 300ml of water to a litre of biodiesel. The biodiesel floats on top of the water. As you mix, the lye, soaps and ethanol are becoming dissolved in the water, which makes it look cloudy. This is due to the fact that water is better at dissolving the impurities than the biodiesel: it is a more polar solvent. The mixing process needs to occur carefully as the soap is an emulsifier and bounds the water and the biodiesel when mixed violently. From the top the biodiesel can be removed carefully and then the washing process can be repeated. As more and more soap is removed from the biodiesel, it becomes less able to bind water molecules, which means that we are using water to dry the fuel. After the washing it is still necessary to give the fuel a final filtration, it removes particles and traps of water. This can be done with a J-cloth or a similar tightly-woven cloth. Normal biodiesel has 1200-1500 ppm (parts per million) of water. Any water above this level will damage your engine.
As water is undesirable in the finished product the biodiesel needs to be dried and filtered so that only the purest biodiesel remains.
|
|