Currently, we are targeting evidence synthesis in the field of Cystic Fibrosis (CF), but we will expand into other areas of Paediatrics as our group grows.
We work closely with the Cochrane CF and Genetic Disorders group of which Professor Alan Smyth is the co-ordiinating editor. Our priority setting work in CF helps to ensure that Cochrane review titles are those which are important to the CF community. Go to the website to see a list of priority titles.

What are we doing about
1. Assessing the evidence base to inform decision making in clinical care
A systematic review attempts to identify, appraise and synthesise all the empirical evidence that meets pre-specified eligibility criteria to answer a given research question.
To do this we use explicit methods aimed at minimising bias, in order to produce more reliable findings that can be used to inform decision making. At present the following Cochrane systematic reviews are being undertaken within Evidence Based Child Health:
- Antibiotic adjuvant therapy for pulmonary infection
- Antibiotic strategies for eradicating Pseudomonas aeruginosa
- Therapies for preventing recurrence of Pseudomonas aeruginosa in people with cystic fibrosis
- Digital technology for monitoring adherence to inhaled therapies and for predicting and intervening early in exacerbations
- Short-acting inhaled bronchodilators in CF
- Dosing regimens for pancreatic enzyme replacement therapy (PERT) in CF
Members of the group are also authors on other reviews which are going through the routine update cycle, including percutaneous lines for intravenous antibiotics in cystic fibrosis, intravenous antibiotics in cystic fibrosis, and long-acting inhaled bronchodilators in cystic fibrosis.
Prof Alan Smyth is coordinating editor of the Cochrane Cystic Fibrosis and Genetic Disorders Group.
2. Identifying evidence gaps in treatment decisions in CF
3. Shaping the future research agenda in partnership with the CF community
We have carried out a programme of research, in partnership with the CF community, to try and identify priority areas for research and clinical trials that may address them.
James Lind Alliance Priority Setting Partnership
In 2016-17 we undertook a James Lind Alliance Priority Setting Partnership (JLA PSP) in CF. This brought together patients, families, healthcare professionals and commissioners to agree shared priorities for research.
Top 10 priorities for clinical research in CF
Here are our final top ten questions as agreed by the CF community:
- What are the effective ways of simplifying the treatment burden of people with CF?
- How can we relieve gastro-intestinal symptoms, such as stomach pain, bloating and nausea?
- What is the best treatment for non-tuberculous mycobacterium (including when to start and what medication)?
- Which therapies are effective in delaying or preventing progression of lung disease in early life?
- Is there a way of preventing CF related diabetes?
- What effective ways of motivation, support and technologies help people with CF improve and sustain adherence to treatment?
- Can exercise replace chest physiotherapy?
- Which antibiotic combinations and dosing plans should be used for CF exacerbations and should antibiotic combinations be rotated?
- Is there a way of reducing the negative effects of antibiotics e.g. resistance risk and adverse symptoms in people with CF?
- What is the best way of eradicating Pseudomonas aeruginosa?
James Lind CF 2
We then took this work further by looking at four of the top ten priorities, which were broad in their nature, in more depth. We wanted to find out what the CF community understood by the priorities and at the same time to try and identify more specific questions that could be answered by clinical research.
We reached out to the whole CF community via online surveys and focus groups. We also made use of videoconferencing so that we could include as many people as possible without cross-infection risks.
The four questions we focussed on are:
- What are the effective ways of simplifying the treatment burden of people with CF?
- How can we relieve gastro-intestinal symptoms, such as stomach pain, bloating and nausea?
- What effective ways of motivation, support and technologies help people with CF improve and sustain adherence to treatment?
- Can exercise replace chest physiotherapy?
IMPACT
The results of this programme of work has led to a number of outputs:
- NIHR – HTA put out a call for research proposals which was based on 7 of our top ten priorities
- The top ten priorities fed into the Cochrane priority titles list
- Primary research (GIFT-CF) looking at imaging of GI function.
- Collaborations with other research teams
- Publications relating to this programme of work
Read the CF Trust article "You said, we did" article to find out more about the impact of the JLA PSP.
James Lind Alliance CF Priority Refresh
We have just completed a refresh of the original top ten priorities, in conjunction with the CF Trust and are planning a programme of work to look at how the current priorities differ.
Here are the final refreshed top ten questions agreed by the CF community;
- What options are available for those not able to take current CFTR modulators (including rarer mutations, not eligible and unable to tolerate)?
- What is the best way to diagnose lung infection when there is no sputum e.g. children and those on modulators?
- How can we relieve gastro-intestinal symptoms such as stomach pain, bloating, and nausea?
- How do we manage an ageing population with CF?
- Is there a way of reducing the negative effects of antibiotics e.g.resistance risk and adverse symptoms in people with CF?
- What are the long-term effects of medications (including CFTR modulators) in CF?
- What are the effects of modulators on systems outside the lungs such as pancreatic function, liver disease, gastro-intestinal, bone density etc?
- What are the effective ways of simplifying the treatment burden of people with CF?
- Can genetic therapies (such as gene editing, stem cell and mRNA technology) be used as a treatment for CF?
- Is there a way of preventing CF related diabetes (CFRD) in people with CF?
4. Understanding gut symptoms in CF
Gut investigations are currently invasive or expose patients to radiation and there is little objective evidence on the mechanisms of gut disease in CF. We aim to use Magnetic Resonance Imaging to assess the gut function and transit in CF. Currently we have completed recruitment for our observational study of people with CF and controls. We are currently open to recruitment for a CFTR modulator cross-over trial.
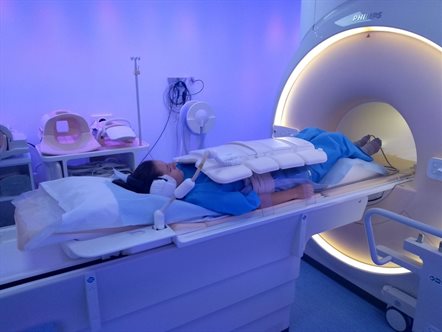
5. A comprehensive approach to the relief of digestive symptoms in CF (CARDS-CF Trial)
Gut symptoms such a tummy pain and bloating are common for people with CF. Our previous research with the CF community told us that 2 in every 3 people with CF miss school or work because of them. The aim of CARDS-CF is to develop a daily scoring system, called a patient reported outcome measure, that can be used in the future by researchers in clinical trials to better assess gut symptoms and the impact that they have on daily life. This will be the first of its kind in CF research.
The study will last up to 2 years and is open to people with CF age 12 years and over. We are currently recruiting for the online focus group and interviews.
For more information the following information sheets are available for download:
Coming soon! CFTR modulators such as Kaftrio are having huge benefits on people with CF lung health, but has it made any difference to gut symptoms? Soon we’ll be launching a new survey open to all people with CF, friends and families and health professionals to find out what gut symptoms are like since starting modulators. We want to hear from people who are not on modulators as well to understand what the similarities and differences in these symptoms are.
Gut Symptom Survey Information Sheet
Current projects
Key publications
Smyth AR, Smith SJ, Rowbotham NJ. Infection prevention and control in cystic fibrosis: One size fits all The argument against. Paediatric Respiratory Reviews 2020;36:94-96. doi: https://doi.org/10.1016/j.prrv.2019.08.001
Langton Hewer SC, Smyth AR, Brown M, et al. Intravenous versus oral antibiotics for eradication of Pseudomonas aeruginosa in cystic fibrosis (TORPEDO-CF): a randomised controlled trial. Lancet Respir Med 2020;8:975-86
Smith S, Rowbotham N, Davies G, et al. How can we relieve gastrointestinal symptoms in people with cystic fibrosis? An international qualitative survey. BMJ Open Respiratory Research 2020;7(1):e000614. doi: 10.1136/bmjresp-2020-000614
Calthorpe RJ, Smith SJ, Rowbotham NJ, et al. What effective ways of motivation, support and technologies help people with cystic fibrosis improve and sustain adherence to treatment? BMJ Open Respiratory Research 2020;7(1):e000601. doi: 10.1136/bmjresp-2020-000601
Ng C, Dellschaft NS, Hoad CL, et al. Postprandial changes in gastrointestinal function and transit in cystic fibrosis assessed by Magnetic Resonance Imaging [published online ahead of print, 2020 Jun 16]. J Cyst Fibros. 2020;S1569-1993(20)30733-5. doi:10.1016/j.jcf.2020.06.004
Smyth AR, Rawlinson C, Jenkins G Preprint servers: a ‘rush to publish’ or ‘just in time delivery’ for science?Thorax Published Online First: 20 April 2020. doi: 10.1136/thoraxjnl-2020-214937
Kalaitzis, I.S., N.J. Rowbotham, S.J. Smith, and A.R. Smyth, Do current clinical trials in cystic fibrosis match the priorities of patients and clinicans? A systematic review. Journal of Cystic Fibrosis., 2019. 19(1): p. 26-33.
Ojha S, Szatkowski L, Sinha R, et al Rojiroti microfinance and child nutrition: a cluster randomised trialArchives of Disease in Childhood Published Online First: 10 October 2019. doi: 10.1136/archdischild-2018-316471
Rowbotham NJ, Smith SJ, Elliott ZC, Leighton PA, Rayner OC, Morley R, et al. Adapting the James Lind Alliance priority setting process to better support patient participation: an example from cystic fibrosis. Research Involvement and Engagement. 2019;5(1):24.
Gichuru W, Ojha S, Smith S, et al. Is microfinance associated with changes in women's well-being and children's nutrition? A systematic review and meta-analysis. BMJ Open 2019;9:e023658
Rowbotham NJ,Smith S, Prayle AP, et al. Gaps in the evidence for treatment decisions in cystic fibrosis: a systematic review. Thorax 2019;74(3):229-36 doi:10.1136/thoraxjnl-2017-210858
Cochrane library
Cochrane CF and Genetic Disorders webpage