Current projects
Chronic Kidney Disease (CKD)
Renal Risk in Derby (RRID) Study
In 2008 we commenced a long-term study of 1741 people with CKD, most of who have not previously seen a kidney consultant. Our initial analysis showed that the majority of people were at relatively low risk for complications and only 6% required assessment by a kidney consultant. Although blood pressure control was good overall, up to a third of the people studied would benefit from better blood pressure control.The study also identified the potential value of some new markers of prognosis including arterial stiffness, skin autofluorescence, serum free light chains and fibroblast growth factor 23. Other analyses have focused on the role of salt intake in contributing to the risks associated with CKD as well as the importance of monitoring protein levels in the urine.We have now completed reassessment of all the participants five years after their first study visit. The information we obtain from these visits should provide valuable insights into the long term complications of CKD. Using this information, we aim to develop methods to predict risk in people with CKD so that those at high risk can be referred early for more intensive treatment, whereas those at low risk will be spared unnecessary referral.
Multiparametric MRI for patients with kidney disease
In collaboration with the Sir Peter Mansfield MRI centre in the School of Astronomy and Physics, we are undertaking a programme of work to advance the use of MRI to study acute and chronic kidney disease. Magnetic resonance imaging (MRI) can show the entire kidney in more detail than other imaging methods, and there is a range of MRI techniques that can assess different aspects of kidney function. Combining several different MRI techniques into a single scan session is called multiparametric MRI. Multiparametric MRI can assess processes that influence or cause kidney disease, such as changes in blood flow, oxygen levels and the degree of scarring (fibrosis). Importantly, MRI scans do not use radiation (unlike X-Rays and CT scans), nor do they need injections of dye (contrast) sometimes needed for other types of scan. This means that MRI scans are completely safe and can be repeated to see whether patients are improving in response to treatment or not.
We aim to utilise these new techniques to visualise pathophysiological changes in the kidneys by measuring blood flow, perfusion and fibrosis. We will correlate these measures with biopsy scores of fibrosis, measured GFR values and long term change in renal function. In parallel, we are also testing novel approaches, such as ex-vivo imaging of kidney tissue, to further establish biological validity of the different MRI measures. Results have the potential to improve assessment of kidney diseases, assess response to treatments and add prognostic information about long term kidney health.
We are also using novel techniques such as 23sodium MRI to characterise kidney patients in ways not previously possible, developing applications in CKD and in dialysis.
The National Unified Renal Translational Research Enterprise (NURTuRE) is the first and only kidney biobank for chronic kidney disease (CKD) and idiopathic nephrotic syndrome (INS), covering England, Scotland and Wales. Biological samples from 3000 patients with CKD and 800 patients with INS are being collected and stored. The biobank will contain anonymised linked clinical data, through the Renal Research Registry, so we can efficiently track kidney health of participants over the next 10 years. Ultimately, we aim to develop new biomarkers to improve our ability to stratify individual patients’ risk. Overall, this will lead to a greater ability to identify patients who will benefit from better earlier diagnosis and person-specific new treatments, leading to better health outcomes.
This project is being jointly delivered through the University of Nottingham, led by Professor Maarten Taal, Professor of Medicine, and the University of Bristol, led by Professor Moin Saleem, Professor of Paediatric Renal Medicine. The research collaboration is under the strategic oversight and management of Kidney Research UK.
More details are available at the NURTuRE project website: www.nurturebiobank.org
Acute Kidney Injury (AKI)
The ARID study (AKI risk in Derby), funded by Kidney Research UK and the British Renal Society, aims to determine the effects of acute kidney injury (AKI) on long term patient outcomes, including the development of CKD.
AKI is very common in hospitalised patients, often occurring when they are unwell from another illness, especially in more vulnerable patients with long term medical conditions. AKI is not a physical injury to the kidney and usually occurs without symptoms. In many cases kidney function improves before patients leave hospital. However, there is relatively little research that follows the progress of their kidney function in the longer term.
We are therefore conducting the ARID study that involves the recruitment of over 1,000 former hospital in-patients, which was achieved by April 2016. We have focused on two groups of people – those who have experienced sudden reductions in kidney function (AKI group) and those who have not (the control or comparator group). Both groups were asked to give blood and urine samples over a three-year period and their general health will be tracked for a further two years. Third year follow ups were completed in January 2019 and we aim to finish five-year follow-ups by 2021. In contrast to most of the current knowledge in the area, the ARID study is prospective and includes a control group, features which differentiate from many other studies of the long term outcomes of AKI. Ultimately, we aim to develop strategies to identify patients who are at higher risk at a much earlier stage, which in turn will enable future work to determine more effective treatments.
Tackling AKI: a multicenter quality improvement project
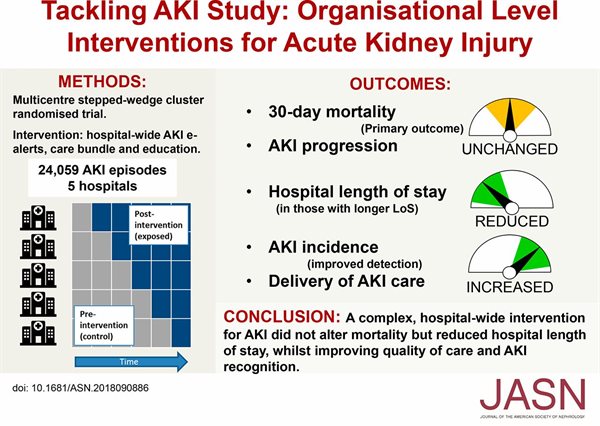
Tackling AKI was a multicentre pragmatic clinical study that was funded by the Health Foundation. The study commenced in 2015 and finished in 2017. The study evaluated the effectiveness of a package of measures to reduce harm associated with AKI, which was introduced across five UK hospitals. The interventions consisted of electronic AKI detection and alerting (using the NHS England algorithm), hospital wide education and an AKI care bundle, with their implementation supported by a dedicated change management strategy. Their aim was to increase recognition of AKI, enhance awareness and understanding and drive improvements in basic elements of care, with an ultimate goal of improving patient outcomes.
The clinical trial studied around 24,000 cases of acute kidney injury, half of which had standard care and half had the package of interventions (consisting of an electronic alert within hospital IT systems, an AKI care-bundle, and an educational programme). The results (see graphical abstract) show that the overall the mortality rate at 30 days after onset was the same in both groups, but significantly, hospital length of stay was reduced, the duration of AKI episodes was shorter, and there was also an increase in AKI incidence explained by improved diagnosis. The interventions also led to improvements in several metrics related to AKI care, including AKI recognition, best use of medication and fluid assessment by clinicians. Together with previous studies, these results show that strategies to improve the systematic delivery of supportive AKI care can lead to improvements in patient outcomes. Furthermore, the reduction in hospital length of stay seen in this study could translate into a significant health economic benefit.
We have published resources on the KQuip website to share materials that were generated from the Tackling AKI study. These materials include study documents and published results, but in the main are the materials that were generated by the project teams at each of the five participating centres. It also includes reflections on ‘lessons learnt’ on key aspects of implementing the intervention on a hospital wide basis.
Contrast Enhanced Ultrasound in AKI
The kidney has an extremely high blood flow, as a quarter of the blood that is pumped by the heart goes to the kidneys. Some of this blood flow is needed to keep the kidneys healthy by delivering oxygen and nutrients; this delivery of the blood to the kidney is called ‘perfusion’. We know that many forms of kidney disease involve a fall in kidney perfusion. In particular, reduced kidney perfusion is a common cause of a sudden reduction in kidney function, called Acute Kidney Injury (AKI).
We are undertaking a research project to test whether a new method of assessing kidney perfusion can improve care for people with AKI. This project is supported by a grant from Kidney Research UK. There are two stages to this project. In the first stage, we will test whether Contrast Enhanced Ultrasound (CEUS) can accurately measure renal perfusion. We will do that by comparing it with the gold standard method using MRI scanning (ASL-MRI) in healthy volunteers and people with chronic kidney disease (CKD). In the second stage, we will perform CEUS in patients who are in hospital and have severe AKI. We aim to find out if this new type of ultrasound scan (CEUS) is a reasonable method to measure kidney perfusion and secondly, if CEUS is useful in patients with AKI. Results from this research will pave the way for the future use of CEUS for the care of patients with kidney disease.
Dialysis treatment
MRI to assess the effects of dialysis
Dialysis treatment is life-saving for people with kidney failure but may also be associated with severe complications affecting the heart, arteries, brain, bones and muscles as well as a risk of infection.
We have completed a ground-breaking study in which we performed functional cardiac MRI during dialysis for the first time. Results have given us unprecedented information about how dialysis acutely affects cardiac structure, function and perfusion. The results for this have been published in a leading nephrology journal: https://jasn.asnjournals.org/content/28/4/1269
We have now completed a follow up study in which intra-dialytic MRI was used to concurrently study the impact of dialysis on the heart, brain and kidneys and the mechanisms by which carefully regulated cooling of the dialysate lessens adverse effects. We hope these results will be available by the end of 2020.
Skin Autofluorescence in dialysis patients
Advanced Glycation End-products (AGEs) are compounds that form naturally in our bodies from the chemical reaction of sugars with proteins. They are also found naturally in foods, and the content of AGEs in foods is increased by cooking methods such as frying, roasting and grilling. If the concentration of AGEs in our bloodstream becomes excessive, they will eventually accumulate in our skin and blood vessels causing damage to almost every tissue and organ in our bodies. Due to their properties to absorb specific wavelengths of light, AGEs can be measured by skin autofluorescence (SAF), a technique which involves placing the forearm on a piece of equipment that shines light on the skin and measures the amount of light that is reflected back. This allows us to measure the concentration of AGEs in the skin.
We are undertaking a series of studies to investigate the relationships between SAF levels, food intake of AGEs and measures of nutritional status, as well as the link between SAF levels, undernutrition and the risk of death. In addition, we aim to explore the factors related to changes in SAF levels over 1 year. So far, the results from this study have given us a better understanding of the associations of AGEs in dialysis patients. We hope that further research will provide new information about how we can change SAF levels, possibly by changes in diet or nutrition.
This is a collaborative project between The Centre for Kidney Research and Innovation and the Institute for Innovation in Sustainable Engineering from the University of Derby.
By working together, we aim to develop technologies to improve patient monitoring during dialysis that will inform and feed refinements in the delivery of dialysis treatments that will ultimately improve patient outcomes. This three year programme of work will develop models to process continuous physiological data in real time, to predict changes of haemodynamic stress prior to their occurrence and ultimately lead to feedback systems involving dialysis based interventions.
Comparison of AV Fistula Cannulation Techniques
Haemodialysis is a life-saving treatment for people who have complete kidney failure, as it replaces the work that the kidneys normally do. It requires special access to the circulation, and this is most commonly done using an arteriovenous fistula or ‘fistula’. A fistula is a special vein created by an operation requiring needles to be inserted for each haemodialysis treatment. This can cause complications and can be painful and stressful for haemodialysis patients.
There are two techniques for putting needles into the fistula, button hole and rope ladder, but currently we do not know which is better at preserving the fistula function and reducing other complications. As part of an NIHR doctoral fellowship, we explore previous studies that have examined patients’ experiences of needle insertion for haemodialysis, as well as designing a questionnaire to measure patients' experiences of needling. In addition, the project will also involve running an initial (feasibility) study to test the large trial design. We aim to explore how to measure patient experience of the two techniques and examine whether a large study can accurately compare the two needle insertion methods. We hope this will inform us which needling technique is best to use.
Chronic kidney disease and dialysis can lead to frequent hospital admissions with reduced patient well-being and quality of life. PREHAB is a research project, funded by a KRUK allied healthcare professional fellowship, which investigates whether regular exercise, good nutrition, and more detailed education improves patient well-being and quality of life.
Patients expected to start dialysis within the next 12 months will be invited to take part, and randomly allocated to a 10-week exercise and education programme, and additional education on starting dialysis, or routine care.
Tests to measure muscle strength, exercise tolerance, appetite, and quality of life, will assess whether the exercise and education programme is more beneficial than standard care. Overall, we hope this will improve the quality of life of patients starting dialysis.
Muscle perfusion during haemodialysis
Functionally significant skeletal muscle wasting in haemodialysis patients is highly prevalent, progressive and predisposes this vulnerable patient population to increased risk of complications, poor quality of life, frailty and premature death. The process of muscle wasting is multifactorial and includes changes in amino acid and lipoprotein metabolism, the effects of inflammation, anorexia and endocrine dysfunction, altered muscle intracellular signaling, and defective myocyte regeneration. In addition, previous preliminary studies have suggested that the process of haemodialysis itself may be associated with muscle catabolism.
Building on previous work, in which we’ve shown that haemodialysis can affect perfusion (blood flow) in different organs, including the heart and the brain, we are now undertaking pilot studies to examine the effect of haemodialysis on muscle perfusion. To do so, we are collaborating with the Clinical, Metabolic and Molecular Physiology group and are using contrast enhanced ultrasound (CEUS) during dialysis to determine whether femoral artery blood flow and thigh muscle perfusion change.