Surface Plasmon Resonance (SPR)
Study molecular interactions and reaction kinetics using SPR
SPR at a glance
Surface Plasmon Resonance (SPR) instruments are optical biosensors that allow sensitive detection of molecular interactions in real time. It is a label free technique thus eliminates the risk of label associated interference of the interaction being studied. SPR is typically used for measuring binding constants of molecular interactions (drug-protein, hormone-protein, protein-protein, nucleic acid-protein, carbohydrate-protein and lipid-protein interactions). It is also used in materials characterisation i.e. characterization of polymer layers and abiotic-biotic interactions.
Capabilities
- Molecular interaction studies (kinetics, affinity, specificity, strength, concentration etc.)
- Analyte identification studies (bacteria, viruses, hormones, immunoglobulins etc.)
- Protein activity and stability analysis
- Epitope mapping
- Characterization of mutant proteins
- Study of layer-by-layer self-assembly
Typical Applications
- Clinical studies i.e. Identification of binding partners to targets by linking SPR to mass spectrometry (proteomics) which can be carried out without need for purification of complex mixtures such as clinical material
- Academic research i.e. life sciences, materials science
- Industry research i.e. pharmaceutical drug discovery (mechanism of action, hit validation and characterisation, lead optimisation), materials characterisation
Image 1: Illustration of the Basic Principle of an SPR Measurement
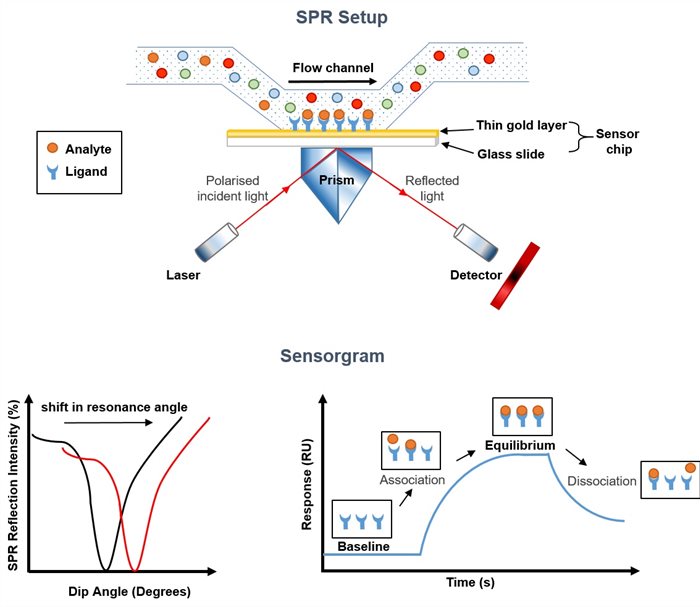
The underlying principle of an SPR measurement is to attach (immobilize) a molecule termed ligand onto a sensor chip surface and measure the change in refractive index of the surface when another molecule (interaction partner called analyte) binds to it. In a standard SPR set up, shown above, a beam of incident polarized monochromatic light is shone through a prism at a thin-layer of gold coating one surface of a glass sensor chip. The prism causes the light to be reflected at the gold-coated surface. At a particular angle of incidence, absorption of some of the light by the electrons in the gold excites charged density waves, called "surface plasmons", which propagate along the metal surface. Plasmon resonance leads to a reduction in the intensity of the reflected light. The angle at which the intensity of refracted light is most significantly reduced (resonance angle) depends not only on the gold layer but also occurs as a function of the refractive index of the medium just above the gold surface i.e. the buffer solution. SPR is thus highly sensitive to changes in the environment close to the gold-aqueous solution interface. A change in the refraction index at the surface of the sensor (due to for example analyte binding or dissociation occurring near the surface) may hence be monitored as a shift in the resonance angle and is recorded as a sensorgram.
Figure courtesy of Marion J. Limo, ISAC, School of Pharmacy, University of Nottingham
Image 2: SPR Kinetic Study of Interaction between Glycopolymers and Lectin
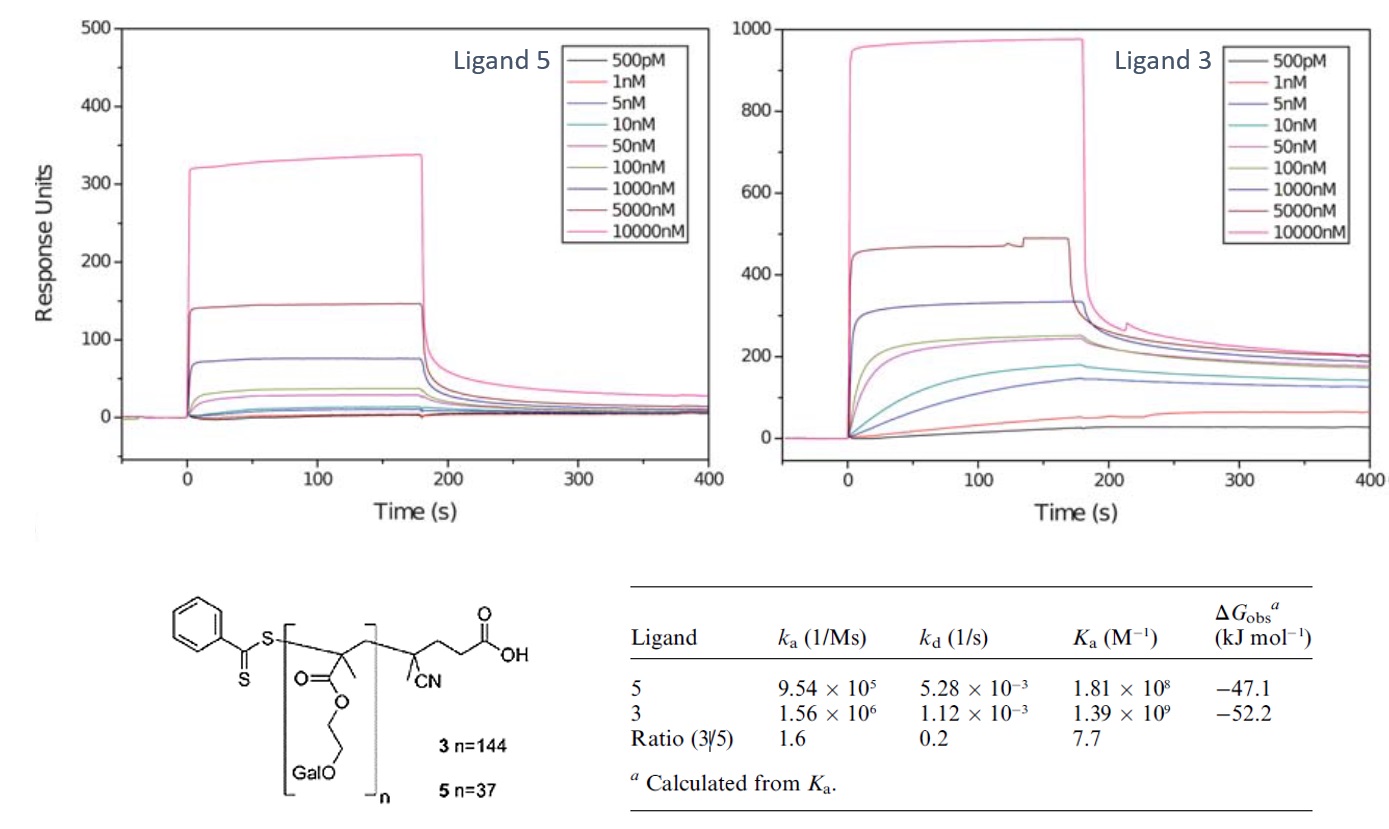
Interaction between carbohydrates and carbohydrate-binding proteins known as lectins has been of interest as a targeting mechanism for drug-delivery because of their known interaction specificity. This interaction is however, weak compared to others in nature but its avidity is ehanced by multivalency. The interaction of two synthesised β-D-galactose bearing polyvalent glycopolymers (ligand 5 and ligand 3) with plant lectin Ricinus communis agglutinin 120 (RCA120) was investigated using a Biacore 3000 SPR. Complex polyvalent glycopolymers with defined valency can be synthesised using relatively facile chemistry. The aim of the study was to investigate the avidity of the glycopolymers for lectins towards the development of drug-delivery vehicles. RCA120 was immobilised on a CM5 sensor chip and the interaction of the two glycopolymers studied at different concentrations (500 pM to 10 µM) as shown in the sensorgrams. The data was fitted with the BIAcore software using a single sites (1 : 1 Langmuir Binding) model. Fitting with highest concentrations and some low concentrations were removed as binding was either too rapid or too weak to enable accurate fitting. Derived kinetic and thermodynamic parameters are shown in the table. Ligand 3 had a much higher avidity for RCA120 binding in comparison to ligand 5, evident from the 5 fold higher kd of ligand 5.
Image adapted from Sebastian G. Spain and Neil R Cameron. Polym. Chem., 2011, 2, 1552-1560.
Our SPR Facilities
The Biacore T200 (Cytivia) is a highly sensitive and fully automated surface plasmon resonance instrument with high-throughput capabilities (up to 384 samples per run) providing high quality molecular interaction data in real time with ultra-low concentrations (fm to mm range). Its applications are wide and include kinetics, affinity, specificity, concentration, immunogenicity, epitope binding, and transition-state thermodynamics. It can run in variable temperatures (4°C to 45°C) and has a refractive index of 1.33 to 1.4. It can detect interactions of a wide range of masses up to lipids, cells and virus interactions.