Gene Regulation and RNA Biology
The Gene Regulation and RNA Biology laboratory interegrates the activities of research teams across two research divisions in the School of Pharmacy.
Division of Biomolecular Science and Medicinal Chemistry:
Dr Sebastiaan Winkler: RNA degradation, CcR-Not complex, nuclease inhibitors
Dr Anna Piccinini: Extracellular matrix regulation of miRNA biogenesis
Prof David Heery: Nuclear receptors, Histone modifications, KAT6A complexes
Dr Hilary Collins: Senior Scientific Officer
Division of Molecular Theraputics and Formulation
Dr Catherine Jopling: miRNA biogenesis; regulation of HCV by miR122
Dr Keith Spriggs: EGFR expression in hypoxic tumours; natural products as anti-cancer agents, bioinformatics
Dr Cornelia de Moor: Poly(A) metabolism in gene expression and as a drug target
The interests of this group are in investigating molecular and cellular mechanisms of the regulation of gene expression in eukaryotic cells, with a focus on the dysfunction of these processes in human disease. We study the function of protein complexes that control many aspects of gene regulation, from chromatin modification and epigenetic regulators through to transcriptional control, mRNA processing and turnover, miRNA regulators and translational control.
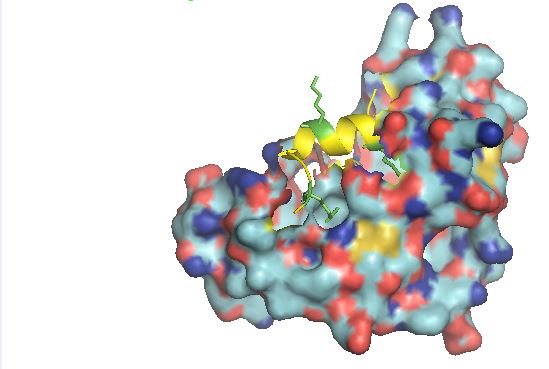
Caption: Crystal structure of the KAT6A/MOZ Double PHD Finger domain in complex with the Histone H3 tail acetylated at Lysine 14.
We share expertise in a range of molecular techniques including protein structure/function analyses, cellular models of physiology and disease, protein-protein and protein-nucleic acid interactions, confocal microscopy, transcriptomics, chIP-Seq, RTqPCR, CRISPR CAS9 genome editing. We have a vibrant team of international postgraduate researchers (currently 18 nationalities) and members of the GRRB laboratory have extensive collaborative networks both locally and internationally. Currently funded projects are supported by BBSRC, Breast Cancer Now, Arthritis UK, Prostate Cancer UK, Academy of Medical Sciences.
Recent publication highlights include:
Recent publication highlights include:
- Airhihen, B. Pavanello, L., Jadhav, G.J., Fischer, P.M., and Winkler, G.S. (2019) 1-Hydroxy-xanthine derivatives inhibit the Caf1 nuclease and Caf1-containing nuclease complexes via Mg2+-dependent binding in the active site. FEBS Open Bio, doi: 10.1002/2211-5463.12605
- GSK3β-SCFFBXW7α mediated phosphorylation and ubiquitination of IRF1 are required for its transcription-dependent turnover. Garvin AJ, Khalaf AHA, Rettino A, Xicluna J, Butler L, Morris JR, Heery DM, Clarke NM.Nucleic Acids Res.2019 Mar 11. pii: gkz163.
- The central region of CNOT1 and CNOT9 stimulates deadenylation by the Ccr4-Not nuclease module.Pavanello L, Hall B, Airhihen B, Winkler GSBiochem J.2018 Nov 9;475(21):3437-3450.
- Predicting puberty in partial androgen insensitivity syndrome: Use of clinical and functional androgen receptor indices. Lek N, Tadokoro-Cuccaro R, Whitchurch JB, Mazumder B, Miles H, Prentice P, Bunch T, Zielińska K, Metzler V, Mongan NP, Heery DM, Hughes IA. EBioMedicine.2018 Oct;36:401-409.
- Mesenchymal Stem Cell-Conditioned Medium Reduces Disease Severity and Immune Responses in Inflammatory Arthritis. Kay AG, Long G, Tyler G, Stefan A, Broadfoot SJ, Piccinini AM, Middleton J, Kehoe O.Sci Rep.2017 Dec 21;7(1):18019
- Expression of NAD(P)H quinone dehydrogenase 1 (NQO1) is increased in the endometrium of women with endometrial cancer and women with polycystic ovary syndrome. Atiomo W, Shafiee MN, Chapman C, Metzler VM, Abouzeid J, Latif A, Chadwick A, Kitson S, Sivalingam VN, Stratford IJ, Rutland CS, Persson JL, Ødum N, Fuentes-Utrilla P, Jeyapalan JN, Heery DM, Crosbie EJ, Mongan NP. Clin Endocrinol (Oxf). 2017 Nov;87(5):557-565.
- Mapping tenascin-C interaction with toll-like receptor 4 reveals a new subset of endogenous inflammatory triggers. Zuliani-Alvarez L, Marzeda AM, Deligne C, Schwenzer A, McCann FE, Marsden BD, Piccinini AM, Midwood KS. Nat Commun.2017 Nov 17;8(1):1595.
- Heterodimers of photoreceptor-specific nuclear receptor (PNR/NR2E3) and peroxisome proliferator-activated receptor-γ (PPARγ) are disrupted by retinal disease-associated mutations. Fulton J, Mazumder B, Whitchurch JB, Monteiro CJ, Collins HM, Chan CM, Clemente MP, Hernandez-Quiles M, Stewart EA, Amoaku WM, Moran PM, Mongan NP, Persson JL, Ali S, Heery DM. Cell Death Dis.2017 Mar 16;8(3):e2677
- Abnormal Clock Gene Expression and Locomotor Activity Rhythms in Two Month-Old Female APPSwe/PS1dE9 Mice. Oyegbami O, Collins HM, Pardon MC, Ebling FJP, Heery DM, Moran PM. Curr Alzheimer Res.2017;14(8):850-860
- H-IPSE is a pathogen-secreted host nucleus infiltrating protein (infiltrin) expressed exclusively by the Schistosoma haematobium egg stage. Pennington LF, Alouffi A, Mbanefo EC, Ray D, Heery DM, Jardetzky TS, Hsieh MH, Falcone FH. Infect Immun. 2017 Sep 18. pii: IAI.00301-17.